Bogotá, D.C., Colombia, December 8, 2014 – Marca País, Procolombia’s division in charge of managing Colombia’s country brand abroad, has signed an alliance with Interventional Concepts, a leading clinical development firm in Miami, FL USA with an office in Colombia and focused on conducting clinical trials in Colombia (www.interventionalconcepts.net), to work together to position Colombia as the premier destination in Latin America to conduct clinical trials. Colombia is able to provide the biopharma and medical device industries with a high enrollment rate, regulatory efficiency by the Instituto Nacional de Vigilancia de Medicamentos (INVIMA), competitive costs, highly qualified and motivated bilingual investigators and boasting an ideal geographical location with fast and direct airline connectivity from the major centers of innovation in the U.S. and Europe.
Clinical trials have been conducted in Colombia for over 25 years and is, together with Mexico, Argentina, Brazil and Peru, one of the best destinations for pharmaceutical clinical trials. However, the majority of the country’s clinical research sites don’t have a direct contact with foreign manufacturers, specially with startup companies that need to conduct early stage (phase I) trials for new medical devices and new pharmaceutical molecules. A large number of clinical trials are sponsored by companies that discover or manufacture such medical products so that they can be approved for commercialization by the regulatory bodies in the markets where they seek to conduct business.
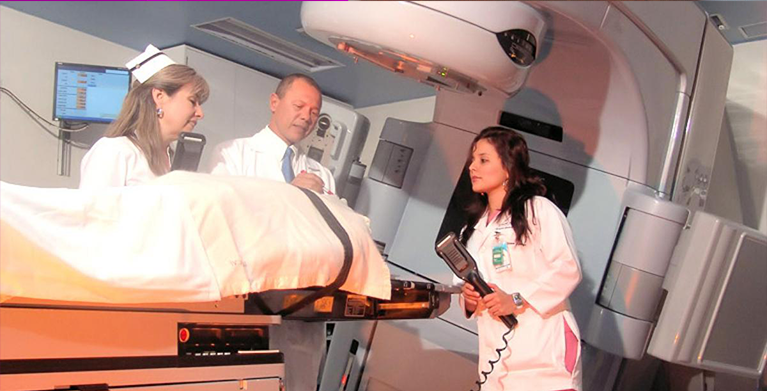
Thanks to this initiative, the medical innovation industry in the U.S. and Europe will have access to over 120 Good Clinical Practices-certified INVIMA research centers in Colombia with over 25 years of experience conducting clinical trials with comparable standards of quality demanded in the U.S. and Europe at very competitive prices. Colombia is world-recognized as a leader and pioneer in health services, and it has expert principal investigators on trials related to cardiology, oncology, diabetes, infectious diseases, vaccines, ophthalmology, endocrinology, hematology, gynecology, among other therapeutic areas.
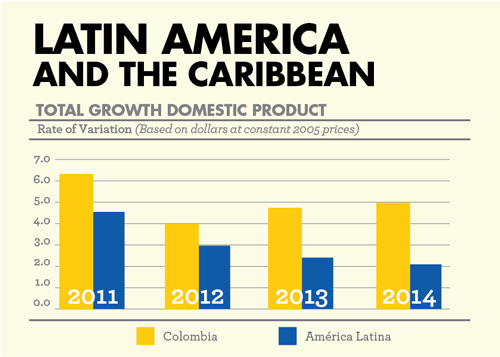
Colombia leads the Latin American ranking of the best clinics and hospitals in the region, and is rapidly emerging as a leader in conducting clinical trials. Based on figures from Colombia’s Ministry of Commerce, Industry and Tourism, in 2013, the country sold over $216 million in health services to foreign medical tourists; a 61% increase over the previous year. This alliance benefits Colombia because it will increase its health service exports, will bring innovation to the country and due to the knowledge transfer that will take place between foreign and local investigators, it will elevate the professional level of the physicians who are part of clinical research centers in Colombia.
From 1995 to 2013, 738 clinical trials have been conducted in Colombia, out of which 90% were sponsored by the pharmaceutical industry and have been related to chronic diseases such as cancer, cardiovascular and degenerative diseases. Colombia has 63 INVIMA-Good Clinical Practices-certified institutional ethics committees. Based on 2012 data, after Brazil, Mexico, Argentina and Chile, Colombia is the sixth largest recipient of clinical research in the Latin America, contributing 9% of all the trials conducted in the region; this creates an opportunity for development and growth of its clinical research activity. Colombia offers benefits to any biopharma or medical device manufacturer in the U.S. and Europe; it has a sizable population —approximately 45 million—, an adequate health infrastructure and highly qualified health professionals. Furthermore, Colombia’s greatest strengths could be its strict regulatory system. INVIMA has established strict requirements to ensure patient safety and quality of clinical research. INVIMA certifies each site in Good Clinical Practices and audits them randomly. Despite INVIMA’s rigor, the country bolsters one of the fastest approval and importation processes.
The Free Trade Agreement between Colombia and the U.S. has allowed the country to become a very desirable commercial partner for foreign manufacturers. According to the World Bank’s Doing Business report, which evaluates local regulations that affect companies in 189 countries and classify them in 10 different areas of business regulations —such as time to incorporate a company, insolvency resolution and transborder commerce—, Colombia is among the easiest countries in which to do business.